ESMO Pocket Guidelines: Lung Cancer – Mesothelioma – Clinical Practice Guidelines
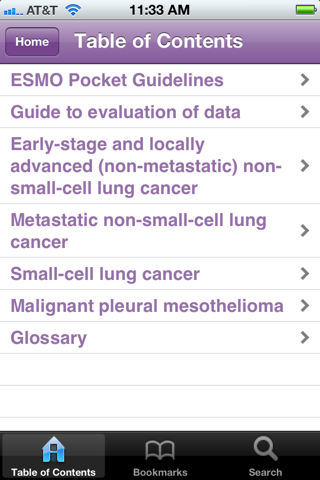
The ESMO Pocket Guidelines: Lung Cancer App provides a concise summary of the fundamental recommendations outlined in the complete ESMO Clinical Practice Guidelines (CPGs) and Consensus Statements on lung cancer. The Guidelines Working Group has brought together all current practical information for the following types of cancer:
•Early-stage and locally advanced (non-metastatic) non-small-cell lung cancer
•Metastatic non-small-cell lung cancer
•Small-cell lung cancer
•Malignant pleural mesothelioma
This App can be used as a quick reference guide to access key content such as diagnostic criteria, staging of disease and risk assessment, treatment plans and follow-up. You will be able to search the text and bookmark specific pages for later research.
The ESMO CPGs are intended to provide you with a set of recommendations for best standards of cancer care, which is based on the findings of evidence-based medicine. ESMO is pleased to release an enhanced and revised set of guidelines that is designed to help oncologists deliver an appropriate quality of care to their patients.
The ESMO Pocket Guidelines: Lung Cancer App is brought to you by Springer Healthcare (www.springerhealthcare.com), a leading global scientific publisher, delivering quality content through innovative information products and services. Please visit www.esmo.org to view the full guidelines and consensus statements.
Disclaimer: Although every effort has been made to ensure that treatment and other information are presented accurately in this publication, the ultimate responsibility rests with the prescribing physician. Neither the publisher nor the Guidelines Working Group can be held responsible for errors or for any consequences arising from the use of information contained herein. For detailed prescribing information on the use of any product or procedure discussed herein, please consult the prescribing information or instructional material issued by the manufacturer.